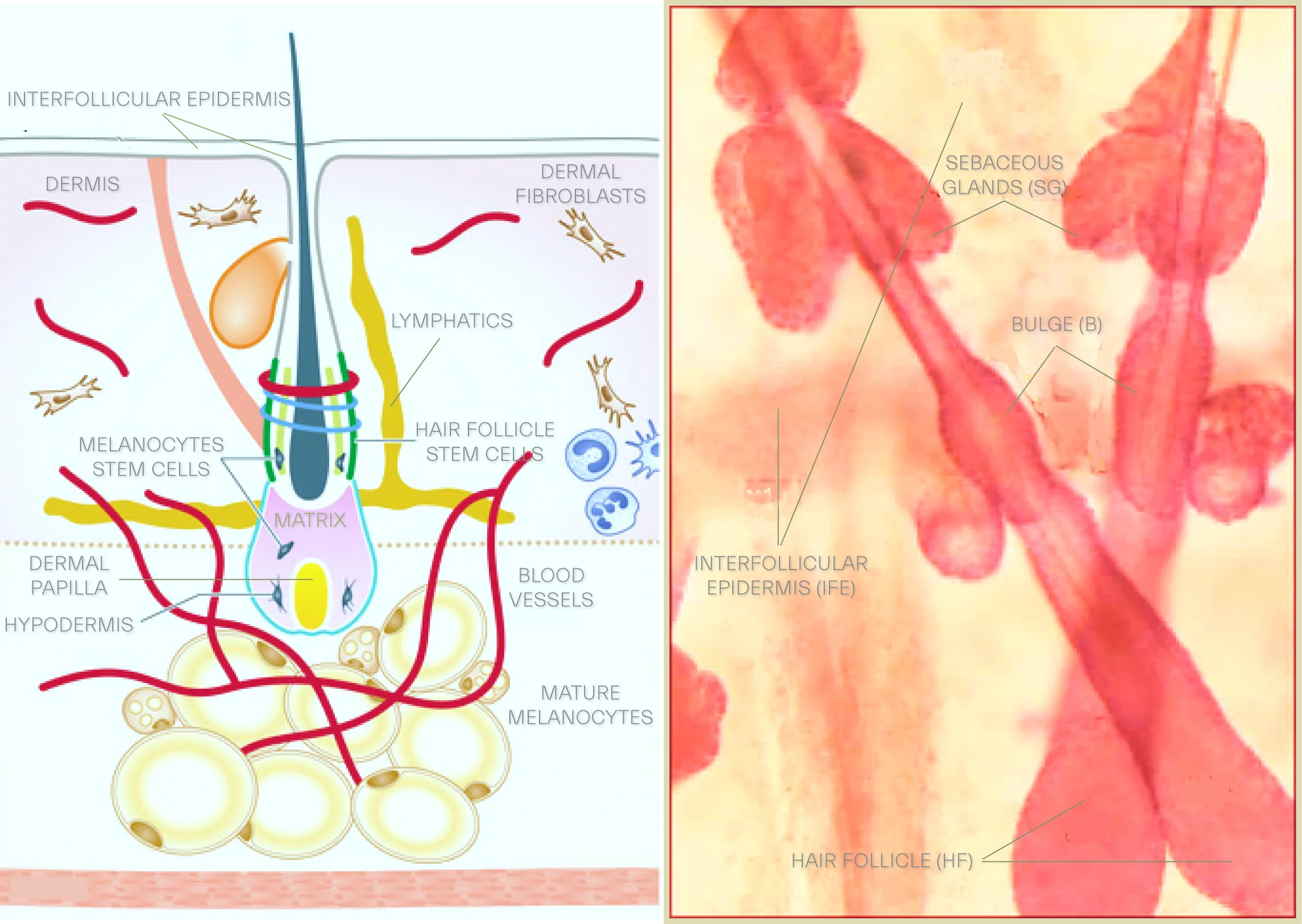
Over the past decade a growing cognizance of sebaceous glands, deeply constitutional to pathophysiology of the skin, delivers a broader and more integrated views of the function of the integument. Sebaceous glands’ output is on a holocrine platform, built upon disintegration of sebocytes where their lipids synthesis into the droplets occurs.
The sebocytes are destined to differentiation thereby they burst and release their fat content and cell debris into the hair follicle. These epithelial glands reside in the dermis, lined with sebocytes, equipped with hormonal receptors and embody skin’s androgen homeostasis, a virtue by which they are properly regarded as an endocrine organ. Sebaceous glands almost always are found associated with hair follicles where these glands secretions end up to with some exceptions such as meibomian glands in eyelids which, without intervention, deliver their content to the surface of the epithelium.
Its production varies with age with an onset at puberty and a nadir after menopause in women or after sixth decade in men. Sebaceous glands differentiation and proliferation is hormonally-controlled and androgen-dependent via binding to androgen nuclear receptors as all enzyme for androgen production and modulation are available in the skin, with 5-alpha-dihydrotestosterone (5α-DHT), the principal player. Men’s skin in general generates more secretions, which could be explained by higher level of androgens. Corticotropin releasing hormone, CRH, is another hormone found in the skin which regulates sebocytes’ function by tempering synthesis of IL-8 in these cells.
Several factors, hormones, nuclear receptors, have been gaining traction for their implication in maturation and differentiation of sebocytes, with insulin as most prominent one. Insulin promote lipid droplet accumulation and sebum build up while further maturation to bursting while epidermal growth factor, EGF, retionoids and vitamin D3 negatively impact it.
SREBPs, sterol regulatory binding proteins, are a family of transcription factors recognized as key elements involved in lipid homeostasis within sebaceous glands, upregulated by liver X receptors, LXRs. SREBP-1c promotes activation of several lipogenic enzymes, namley, stearoly-C0A and Δ-6 desaturase, giving rise to increased proportion of monounsaturated fatty acids in sebaceous glands triglycerides such as sapienic acid. Among vitamins that increase sebaceous production, Vitmain A is known to be essential for sebocyte viability and differentiation albeit their proliferation is mainly affected by retinoids.
Central to the sebaceous glands function is what has been recognized as the secretome, growth factors, hormones, cytokines secreted by SGs before cell disruption, which despite making key contributions to immunemodulation of pilosebaceous milieu even under physiological status are not as yet well studied. One research on SZ95 sebocytes to determine major proteins associated with the secretome found IGF-binding protein 3, IGFBP3, as most abundant protein with regulatory function on IGF bioavailability, a principal modifier of the skin lipogenesis. Epidmeral growth factor, EGF, has been found to induce immune-related genes in sebocytes and bestow upon them a bioactivity they do not possess in its absence. EGF also activates IL-1 signaling which nurtures inflammation, keratinocytes proliferation and their aberrated differentiation, giving rise to comedogenesis.
Sebaceous output contains bioactive molecules whose function is not merely to serve as humectants but labor to protect against environmental extremes by their commitment to UV protection or antimicrobial activity as sebaceous-glands-rich areas of the skin presents distinct immunology from sebaceous-deficient segmentaries. Following differentiation and apoptosis of sebocytes, sebaceous glands products are formed by decomposition of lipid-containing cells, sebocytes, so cell debris, disintegrated mature sebocytes, as well as matrix metaloproteinases, MMP, can be found in its lipids.
Three major lipid classes in human variant are triglycerides and free fatty acids, FFA (58%), wax esters, WE, (26%) and squalene , SQ(12%). Importantly, synthesis of SQ and WE is characteristic of sebaceous gland activity and is found no where else in the body even in epidermal surface lipids excreted from keratinocytes. Moreover, branched chain fatty acids and lipids with specific pattern of unsaturation are also exclusive to the sebaceous products. Highly specific and concentrated in sebaceous outputs, squalene is an unsaturated hydrocarbon and withal a precursor of cholesterol in most human tissues, where this conversion occurs. The triglycerides and FFA make up less than 60 percent of sebaceous glands products while their presence in skin surface lipids reaches to about 40 percent.
Free fatty acids
They form 15-30% and by some other reports 5-40% of sebaceous glands output. The most abundant is sapienic acid (C16 : 1Δ6) and the next in copiousness is sebaleic acid (C18 : 2Δ5,8), both found exclusively in the sebaceous secretions. The Δ6 unsaturated fatty acid, sapienic acid, not seen in any other human tissue or in the sebaceous outputs of other animals. Δ6 desaturase, FADS2, is the very same enzyme which account for conversion of linoleic acid to Gamma-linolenic acid (18:3n-6), GLA, yet it seems its having higher affinity for linoleate justifies the fact that sapienic acid remains sebaceous-specific since linoleic acid undergoes rapid beta-oxidation within the sebocytes. In contrast, FADS1 desaturates dihomolinlenic acid to arachidonic acid (C20: 4n-6), AA.
Role of free fatty acids in comedone formation spawns controversy and various reports encourage cleaving to caution. Sapienic acid, which comprises 25% of total sebocyte-manufactured fatty acids, along with other unsaturated long-chain fatty acids in the sebaceous products such as linoleic acid (18:2, Δ9,12), an ω6 polyunsaturated compound, demonstrates inhibitory effect on staphylococcus aureus, a suggested culprit in chronic inflammatory skin diseases including rosacea, psoriasis and in acne vulgaris via stimulation of toll like receptors 2 (TLR2), the most well described bacterial pathogen in acne is C. acnes.
Another study asserts that sapienic acid arrest the growth of S. aureus without recovery, while along with other research works introduces palmitoleic acid (16:1, 9z) as mammalian most efficient vector against this nuisance. In contrast, unsaturated fatty acids, oleic acid and palmitoleic acid, and not triglycerides or saturated fatty acids, enhance calcium ion influx and promote abnormal keratinazation of follicular epithelium and epidermal hyperplasia, one study explains.
Linoleic acid (C18:2Δ6), LA, is briskly converted its derivatives within sebocytes by presence of a Δ-6 desaturase, product of FADS2 gene and specific to human SG, which also from palmitic aid (C16:0) produces sapienic acid, altered to sebaleic acid upon further desaturation. Δ-6 desaturase also works on other PUFAs, poly unsaturated fatty acids, including alpha linolenic acid (C18:3Δ3), ALA, in human sebaceous glands. Dietary increase in linoleic acid does not lead to a significant change in proinflammatory arachidonic acid (C20:4Δ6), AA, as it is been suggested that less than 1% of linoleic acid transforms into AA. Δ-6 desaturase and the metabolites of its work are indexes of sebocyte differentiation, compromised in certain skin diseases such as acne, posited having lower level of C 16:0/C 16: 1.
Another study also supports antimicrobial activity of stratum corneum lipids against methicilin resistant staphylococcus aureus (MRSA), far greater effective than mupirocin. This product of sebocytes is not found in human diet as only a few plant species produces this fatty acid. In contrast, sebaleic acid, a polyunsaturated fatty acid formed in sebocytes by elongation of sapienic acid, is esteemed pro inflammatory and chemoattractant for neutrophils. Oleic acid, an ω9 monounsaturated fatty acid have been shown having antibacterial properties. Not as abundant as other fatty acids, lauric acid (C12: 0), 1-2% of total fatty acids of the sebacous glands output, shows remarkable therapeutic antibacterial effect against C. Acnes, S. epidermidis and S. aureus, several studies proposed.
Poly unsaturated fatty acids, PUFA, are cateogrized into ω-6, linoleic acid and arachidonic acid, and ω-3 fatty acids, α-linolenic acid, eicosapentaenoic acid, EPA and dicosahexaenoic acid, DHA. Both categories, ω-6 and ω-3, are scarcely found in the sebacous products. While ω-3 fatty acids are, by large, esteemed as anti-inflammatory by plethora of mechanisms, they compete with arachidonic acid for incorporation into the cell membrane and thereby lower proinflammatory moeities, produced by lipoxygenase or cycloxygenase, content of the membrane. The lower level of ω-3 may be accounted for by the fact that ω-3 substrate, linoleic acid undergoes β-oxidation, leaving room for palmitic acid convertion to sapienic acid and sebalic acid by sebaceous desaturase.
Under conditions which do not allow differentiation of sebaceous cells, linoleic acid does not beta-oxidize to two-carbon precursors and stays available to outcompete palmitic acid for access to the ambient desaturase leading to formation of more proinflammatory products such as arachidonic acid. Less differentiated sebocytes possess lower level of linoleic acid in their more complex lipids such as acyl-ceramides or wax esters as if sebaceous machinery once under stress and overpoduction generates less luxariant substances and admittedly, no matter how fine the analogy is the discretion may not be gainsaid.
Not found in significant amount in circulating lipids, GLA, rapidly coverts, by virtue of an elongase, into dihomo gamma linolenic acid (C20:3n-6), DGLA, which more prominently integrates into phospholipid pool of cell membranes to a more measurable extent. DGLA exhibit a constraining impact on platelet aggregation and inflammation through its anti inflammatory metabolites, PGE1 and 15-HETrE, as well as its cometition with arachidonic acid, AA, an initial molecule in cycloxygenase and lipoxygenase cascades. While final fate of GLA spawns controversy as it presents a complex heterogenity in various cells, it appears the equlibrium between the two bioactives, DGLA and AA, which allege final outcome of GLA.
Atopic dermatitis patients have been shown to increase serum level of GLA and DGLA and improve symptoms upon 4-8 weeks dietary supplementation with evening primrose oil, containing more than 10% GLA. One study suggest that in certain human cells such as neutrophils, the transformation of GLA to AA does not occur while some other studies propose this conversion is insignificant in humans, however, cutaneous modulation of GLA to AA has not been investigated. Consumption of GLA with n-3 PUFA such as alpha linolenic acid lean the balance more toward generation of DGLA rather than AA and does not lead to generation of leukotrienes.
As above studies imply, simplistic assertion of inflammatory linoleic acid which infiltrates lay media and some unreviewed medical papers appears to consist on untenable grounds as a spectrum of complexity claims the fate of omega-6 fatty acids in human body and far more critically in sebaceous glands, despite the potentially viable conversion of linoleic acid to arachidonic acid and its bioactive metabolties.
Squalene
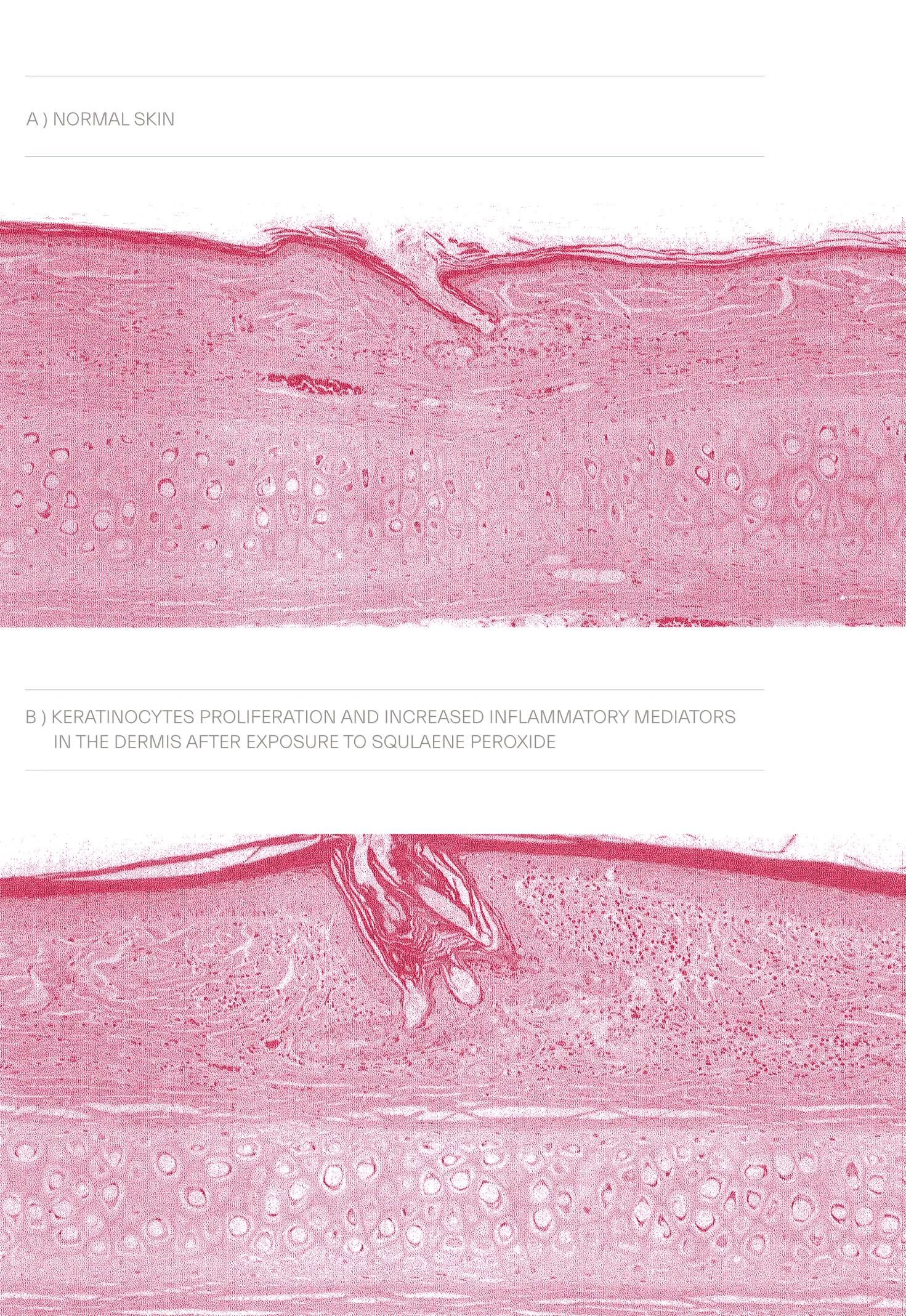
Comprises about 12-20% of sebaceous output, another study suggest 10-14%, squalene, a long unstaturated hydrocrabon chain, is exclusive to sebaceous gland activity, a marker of this gland differentiation and in other tissues converts to cholesterol. Despite its role in UV protection, squalene, amid highly unsaturated skin surface lipids, is more vulnerable to peroxidation and formation of squalene peroxide and in large lipid peroxidation products tend to be proinflammatory, promote progression of comedogenesis and inductive of contact dermatitis.
Peroxidation of squalene may also lead to an inflammatory response in keratinocytes by lipooxygenase activation and upregulation of IL-6 resulting in pilosebaceous duct hyperkeratinization. To counteract the deleterious impact of polar lipids animated by peroxidation, the sebaceous output is limned to unleash a lipophilic antioxidant, vitamin E, α and γ tocopherols, known to be one of its critical constituents.
Wax esters
The second most common lipid of sebaceous glands, more resistent to oxidation and hydrolysis, wax esters make up to 30% of sebaceous content and their biosynthesis is a marker of sebogenesis and sebocyte function and differentiation. DGAT are family of enzymes engaged in sebogenesis, catalazying wax ester formation and triacylglycerol production by DGAT1 and DGAT2 respectively. Esterification of free fatty acids with fatty alcohols lead to formation of these skin oils, of paramount importance in skin barrier function.
Unlinke linoleate, other fatty acids such as stearate, oleate, palmitate and palmitoleate are incoroporated into esterified lipids without further catabolization and oxidation. One study proposes that saturated fatty acids more than unsaturated are incorporated into wax esters with predominance of palmitic acid and that 16-carbon fatty acids comprising 50% of fatty acid content of the sebaceous products.
Linoleic acid is the only fatty acid subjected to beta oxidation and production of acetyl-CoA which upon further metabolism converts into squalene and wax esters. Stearate, oleate, palmitate and palmitoleate are incorporated into more complex lipids without being cleaved to any oxidation or breakdown molecules. Wax esters tend to be generated with saturated fatty acids as palmitic acid is found in higher rate in these sebaceous products whereas oleic acid was found much less and palmitoleic was not detected at all, more likely, incorporated into polar lipids, sphingolipids and phospholipids, for its role in membrane fluidity.
Linoleic acid oxidation is the marker of sebaceous cell differentiation and function and it does not occur under circumstances that prevent sebaceous differentiation. This translates into the fact that linoleic acid is found far less in complex lipids, wax esters, ceramides, in hyperproliferative sebocytes and gives rise to other moieties, with more saturation, whose fate is not well ascertained but still remains a venue to examine for inflammation leak. Overzealous sebaceous glands have been proposed to be depleted in linoleic acid content of their WE, whose synthesis in sebaceous glands is related to exclusive beta oxidation of linoleic acid.
The insight that sebaceous glands FA composition is not fixed and it varies with increased sebogenesis is proved by finding increased sebaleic acid and decreased linoleic acid in acne patients compare to normal controls. Depleted linolenic acid challenges epidermal barrier function and increases vulnerability of comedonal wall to inflammatory mediators and precipitates microcomedone formation. Moreover, hyperproliferative states in keratinocytes may be associated with low LA and ALA as a correlation has been delineated between basal cell carcinoma and squamous cell carcinoma and lower serum level of aforementioned fatty acids.
The change in its composition occurs in two axes, one involves lipid class composition, more WE than CE and the other implicates fatty acid proportions, more Δ6 content in comparison with Δ9. Gravely susceptible to low proportion of linoleate, a remodeling in skin ceramides occurs and follicular epithelium shifts from linoleate-rich acylceramides to sebaceous-fatty-acids acylcermides, which, in turn culminates in hyperkeratosis of follicular epithelium. Squalene increase is suggested as the primary lipid alteration due to sebogenesis in patients with acne and its enhanced ratio in the sebaceous products amounts to severity of acne vulgaris, upregulated 2.2 folds.
Multitude of studies suggest mechanisms by which the skin microbiome affect the sebaceous glands and its constellation of ingredients. Cutibacterium acnes, the bacteria which has been remarked with a causal link to acne lesions, affects sebaceous glands lipogenesis and changes skin surface lipids composition by delivering less complex lipid structures, shifting from triglycerides to free fatty acids and elevated level of desaturated fatty acids as by products of its metabolization.
Cutibacterium acnes affects sebaceous lipogenesis, inflammation and innate immunity at piolosebaceous level. Under anearobic condition C. acnes uses its lipase to catabolize triacylglycerol, amply supplied within sebaceous output, to produce short chain fatty acids, SCFAs, such as propionic acid, isobutyric acid, isovaleric acid, proved to be protective against S. epidermidis biofilm formation.
Some studies provide evidence on how polyphenols and procyanidines may mitigate increase in acid mantle secretion and challenge acne pathology. Another well known manipulator of relationship between its secretion and acne lesions is calcium and vitamin D3. Sebocytes vitamin D receptor has been verified by several lines of study. Indeed, an increase in extracellular calcium and decrease in 1,25(OH)2D3 may account for increase in sebaceous gland volume and lipogenesis.
Several studies in the past thirty years propose that skin in maintaining its barrier to water loss is not dependent to sebocytes as children with almost zero sebaceous products synthesis do not generally show dry skin. Another study in elderly was found no association between dry skin and sebaceous glands activity. Expressing nuclear hormone receptors, sebaceous glands PPAR, peroxisome proliferator-activated receptor, engage in lipid metabolism and inflammation.
Amid PPAR isoforms, PPAR-γ s signifies an instrumental role in sebaceous lipid synthesis induced by oxidative stress, de-escalation of inflammatory mediators and sebocytes proliferation. Drugs which target these nuclear hormone receptors are proposed to be beneficial in treatment of these skin disorders.
Disruption of epidermal barrier function is the primary underlying mechanism known in atopic dermatitis and whether immune dysfunction precede or follow this culprit sparks growing disputes.. Alteration in the epidermal lipids which includes 50% ceramides, 15% FFA, 25% cholesterol and triglycerides, many of which affected by sebaceous lipids and sebaceous glands homeostasis, has been suggested by multitude of studies.
Despite the fact that inflammation, a key modulator of atopic dermatitis, for several decades has been mainly targeted for medical treatment using corticosteroids, restoring epidermal barrier seems to be less injurious, more salutatory and still deserves the challenge of exploration through atopic dermatitis skin care regimens which conform to skin’s original surface lipids.
Enlargement of sebaceous glands has been introduced to be associated with skin aging, in a benign condition known as sebaceous hyperplasia and withal androgenic alopecia. Sebaceous hyperplasia presents itself with small, yellowish papules most commonly on the face, nose, forehead, cheeks are frequently involved, as well as on the body. Oily scalp and how to control sebaceous production on scalp may be a source of concern for younger adults, but in most cases is relieved with aging.
References:
1. Stewart ME, Downings DT. Proportions of Various Straight and Branched Fatty Acid Chain Types in the Sebaceous Wax Esters of Young Children. J Invest Dermatol. 1985 Jun;84(6): 501-3
2. Harder J, Schröder JM, Gläser R. The skin surface as antimicorbial barrier: present concepts and future outlooks. Exp Dermatol.2013 Jan;22(1):1-5
3. Jacobsen E, Billings JK, Frantz RA et al. Age-Related Changes in Sebaceous Wax Ester Secretion Rates in Men and Women. J Invest Dermatol. 1985 Nov;85(5):483-5
4. Fritsch M, Orfanos CE, Zouboulis CC. Sebocytes are the key regulators of androgen homeostasis in human skin. J Invest Dermatol. 2001 May;116(5):793-800
5. Porter AMW. Why do we have apocrine and sebaceous glands? J R Soc Med. 2001 May; 94(5): 236–237
6. Picardo M,Ottaviani M, Camera E. Sebaceous gland lipids.Dermatoendocrinol. 2009 Mar-Apr; 1(2): 68–71
7. Chen WC, Zouboulis CC. Hormones and the pilosebaceous unit. Dermatoendocrinol. 2009 Mar-Apr; 1(2): 81–86
8. Sato T, Shibata F, Koiwai T, et al. Different regulation of lipogenesis in sebocytes and subcutaneous preadipocytes in hamsters in vitro. Biochem Biophys Rep. 2020 Jul; 22: 100761
9. Smith TM, Cong Z, Gilliland KL, et al. Insulin-Like Growth Factor-1 Induces Lipid Production in Human SEB-1 Sebocytes Via Sterol Response Element-Binding Protein-1. J Invest Dermatol. 2006 Jun;126(6):1226-32
10.Zouboulis CC, Xia L, Akamatsu H, t al. The human sebocyte culture model provides new insights into development and management of seborrhoea and acne. Dermatology. 1998;196(1):21-31
11. Dahlhoff M, Fröhlich T, Georg J. Arnold GJ, et al. LC-MS/MS analysis reveals a broad functional spectrum of proteins in the secretome of sebocytes. Exp Dermatol. 2016 Jan;25(1):66-7
12. Törőcsik D, Fazekas F, Póliska S, et al. Epidermal Growth Factor Modulates Palmitic Acid-Induced Inflammatory and Lipid Signaling Pathways in SZ95 Sebocytes. Front Immunol. 2021; 12: 600017
13. Béke G, Dajnoki Z, Kapitány A, et al. Immunotopographical Differences of Human Skin. Front Immunol. 2018; 9: 424
14. Anna Wróbel, Seltmann H, Fimmel S, et al. Differentiation and Apoptosis in Human Immortalized Sebocytes. J Invest Dermatol. 2003 Feb;120(2):175-81
15. Papakonstantinou E, Aletras AJ, Glass E, et al. Matrix Metalloproteinases of Epithelial Origin in Facial Sebacous products of Patients with Acne and their Regulation by Isotretinoin. J Invest Dermatol. 2005 Oct;125(4):673-84
16. Camera E, Ludovici M,Galante M, et al. Comprehensive analysis of the major lipid classes in sebaceous output by rapid resolution high-performance liquid chromatography and electrospray mass spectrometry. J Lipid Res. 2010 Nov; 51(11): 3377–3388
17. Pappas A, Anthonavage M, Gordon JS. Metabolic Fate and Selective Utilization of Major Fatty Acids in Human Sebaceous Gland. J Invest Dermatol. 2002 Jan;118(1):164-71
18. Smith RN, Braue A, George A Varigos GA, et al. The effect of a low glycemic load diet on acne vulgaris and the fatty acid composition of skin surface triglycerides. J Dermatol Sci. 2008 Apr;50(1):41-52
19. Moran JC, Alorabi JA, Horsburgh MJ. Comparative Transcriptomics Reveals Discrete Survival Responses of S. aureus and S. epidermidis to Sapienic Acid. Front Microbiol. 2017; 8: 33
20. Neumann Y, Ohlsen K, Donat S. The effect of skin fatty acids on Staphylococcus aureus. Arch Microbiol. 2015; 197(2): 245–267
21. Subramanian C, Frank MW, Batte JL, et al. Oleate hydratase from Staphylococcus aureus protects against palmitoleic acid, the major antimicrobial fatty acid produced by mammalian skin. J Biol Chem. 2019 Jun 7; 294(23): 9285–9294
22. Katsuta Y, Iida T, Inomata S, et al. Unsaturated Fatty Acids Induce Calcium Influx into Keratinocytes and Cause Abnormal Differentiation of Epidermis.J Invest Dermatol. 2005 May;124(5):1008-13
23. Ge L, Gordon JS, Charleen Hsuan C, et al. Identification of the Δ-6 Desaturase of Human Sebaceous Glands: Expression and Enzyme Activity. J Invest Dermatol. 2003 May;120(5):707-14
24. Rett BS, Whelan J. Increasing dietary linoleic acid does not increase tissue arachidonic acid content in adults consuming Western-type diets: a systematic review. Nutr Metab (Lond). 2011; 8: 36
25. Ottaviani M, Camera E, Picardo M. Lipid Mediators in Acne. Mediators Inflamm. 2010; 2010: 858176
26. Drake DR, Brogden KA, Dawson DV. Thematic Review Series: Skin Lipids. Antimicrobial lipids at the skin surface. J Lipid Res. 2008 Jan;49(1)4-11
27. Cossette C, Patel P, Anumolu JR, et al. Human Neutrophils Convert the Sebaceous-derived Polyunsaturated Fatty Acid Sebaleic Acid to a Potent Granulocyte Chemoattractant. J Biol Chem. 2008 Apr 25; 283(17): 11234–11243
28. Nakatsuji T, Kao MC, Zhang L, et al. Sebaceous Free Fatty Acids Enhance the Innate Immune Defense of Human Sebocytes by Upregulating β-Defensin-2 Expression. J Invest Dermatol. 2010 Apr; 130(4): 985–994
29. Nakatsuji T, Kao MC, Fang JY, et al. Antimicrobial Property of Lauric Acid Against Propionibacterium acnes: Its Therapeutic Potential for Inflammatory Acne Vulgaris. J Invest Dermatol. 2009 Oct; 129(10): 2480–2488
30. Yang D, Pornpattananangkul D, Nakatsuji T, et al. The Antimicrobial Activity of Liposomal Lauric Acids Against Propionibacterium acnes. Biomaterials. 2009 Oct; 30(30): 6035–6040
31. Yeung J, Tourdot BE, Adili R, et al. 12-HETrE, a 12-LOX oxylipin of DGLA, inhibits thrombosis via Gαs signaling in platelets. Arterioscler Thromb Vasc Biol. 2016 Oct; 36(10): 2068–2077
32. Dagmar S, Peter AE, Siegfried B, et al. Gamma-Linolenic Acid Levels Correlate with Clinical Efficacy of Evening Primrose Oil in Patients with Atopic Dermatitis. Adv Ther. 2014; 31(2): 180–188
33. Sergeant S, Rahbar E,Chilton FH. Gamma-linolenic acid, Dihommo-gamma linolenic, Eicosanoids and Inflammatory Processes. Eur J Pharmacol. 2016 Aug 15; 785: 77–86
34. Lee TC, Ivester P, Hester AG, et al. The impact of polyunsaturated fatty acid-based dietary supplements on disease biomarkers in a metabolic syndrome/diabetes population. Lipids Health Dis. 2014; 13: 196
35.Surette ME, Koumenis IL, Edens MB, et al. Inhibition of leukotriene synthesis, pharmacokinetics, and tolerability of a novel dietary fatty acid formulation in healthy adult subjects. Clin Ther. 2003 Mar;25(3):948-71
36. De Luca C, Valacchi G. Surface Lipids as Multifunctional Mediators of Skin Responses to Environmental Stimuli. Mediators Inflamm. 2010; 2010: 321494
37. Tochio T, Tanaka H, Nakata S, et al. Accumulation of lipid peroxide in the content of comedones may be involved in the progression of comedogenesis and inflammatory changes in comedones. J Cosmet Dermatol. 2009 Jun;8(2):152-8
38. Ottaviani M, Alestas T, Flori E, et al. Peroxidated Squalene Induces the Production of Inflammatory Mediators in HaCaT Keratinocytes: A Possible Role in Acne Vulgaris. J Invest Dermatol. 2006 Nov;126(11):2430-7
39. Packer L, Weber SU, Thiele JS. Sebaceous Gland Secretion is a Major Physiologic Route of Vitamin E Delivery to Skin. J Invest Dermatol. 1999 Dec;113(6):1006-10
40. Nikkari T. Comparative chemistry of sebaceous products. J Invest Dermatol. 1974 Mar;62(3):257-67
41. Feussner I, Kühn H, Wasternack C. Do specific linoleate 13-lipoxygenases initiate beta-oxidation? FEBS Lett. 1997 Apr 7;406(1-2):1-5
42. Nordstrom KM, Labows JN, McGinley KJ, et al. Characterization of wax esters, triglycerides, and free fatty acids of follicular casts. J Invest Dermatol. 1986 Jun;86(6):700-5
43. Wertz PW. Lipids and the Permeability and Antimicrobial Barriers of the Skin. J Lipids. 2018; 2018: 5954034
44. Stewart ME, Grahek MO, Cambier LS, et al. Dilutional effect of increased sebaceous gland activity on the proportion of linoleic acid in sebaceous wax esters and in epidermal acylceramides. J Invest Dermatol. 1986 Dec;87(6):733-6
45. Saurat JH. Strategic Targets in Acne: The Comedone Switch in Question. Dermatology. 2015;231(2):105-11
46. Seviiri M, Law MH, Ong JS, et al. Polyunsaturated fatty acid levels and the risk of keratinocyte cancer: A Mendelian Randomisation analysis. Cancer Epidemiol Biomarkers Prev. 2021 Jun 4;cebp.1765.2020
47. Pappas A, Johnsen S, Liu JC, et al. Sebaceous products analysis of individuals with and without acne. Dermatoendocrinol. 2009 May-Jun; 1(3): 157–161
48. Frantz RA, Kinney CK, Downing DT. Variables associated with skin dryness in the elderly. Nurs Res. Mar-Apr 1986;35(2):98-100
49. Inoue T, Miki Y, Kakuo S, et al. Expression of steroidogenic enzymes in human sebaceous glands. J Endocrinol. 2014 Sep;222(3)301-12
50. Bhattacharya N, Sato WJ, Kelly A, et al. Epidermal Lipids: Key Mediators of Atopic Dermatitis Pathogenesis. Trends Mol Med. 2019 Jun; 25(6): 551–562
51. Kim BE, Leung DYM. Significance of Skin Barrier Dysfunction in Atopic Dermatitis. Allergy Asthma Immunol Res. 2018 May; 10(3): 207–215
52. Zouboulis CC. Propionibacterium acnes and Sebaceous Lipogenesis: A Love–Hate Relationship? J Invest Dermatol. 2009 Sep;129(9):2093-6
53. Rozas M, Hart de Ruijter A, Fabrega MJ, et al. From Dysbiosis to Healthy Skin: Major Contributions of Cutibacterium acnes to Skin Homeostasis. Microorganisms. 2021 Mar; 9(3): 628
54. Nakamura K, O’Neill AM, Williams MR, et al. Short chain fatty acids produced by Cutibacterium acnes inhibit biofilm formation by Staphylococcus epidermidis. Sci Rep. 2020; 10: 21237
55. Saric S, Notay M, Sivamani RK. Green Tea and Other Tea Polyphenols: Effects on Sebaceous Production and Acne Vulgaris. Antioxidants (Basel). 2017 Mar; 6(1): 2
56. Zouboulis CC, Seltmann H, Abdel-Naser MB, et al. Effects of Extracellular Calcium and 1,25 dihydroxyvitamin D3 on Sebaceous Gland Cells In vitro and In vivo. Acta Derm Venereol. 2017 Mar 10;97(3):313-320.
Some related questions, found common among teens:
What does excess sebum look like?
Excess sebaceous glands production presents itself with an oily skin
Is sebum a pimple?
The answer is no, sebaceous products are natural skin secretions while a pimple is a disease state manifests with comedones, papules, cysts and nodules
How do you get rid of sebum?
Getting rid of sebaceous output is not favorable as it is protective in multitude of respects but in certain skin disorders, oversynthesis and build up of sebaceous products may be controlled
Is sebum good for your skin?
Today’s medical research understands sebaceous glands products as generally essential and vital to a healthy skin
Is there sebum anywhere on the body besides the skin?
No, but sebaceous products have been identified in other parts of the body
Does sebum contain bacteria?
Certain bacteria resides in sebaceous gland and are associated with its products as the skin possess a natural microbiome that are not generally harmful and by some token symbioticly cohabit, commensal organisms